A recently published Science magazine article details research conducted at the University of Louisiana at Lafayette that could protect chimpanzees and gorillas in the wild from the deadly Ebola virus.
It also indicates, however, that great apes may never benefit from the encouraging vaccine trial.
The online article is based on the research of Dr. Peter Walsh, a disease ecologist at the University of Cambridge in the United Kingdom, and scientists at UL Lafayette’s New Iberia Research Center. They tested a vaccine that is similar to a rabies vaccine that has been successful in immunizing wild animals such as foxes and coyotes.
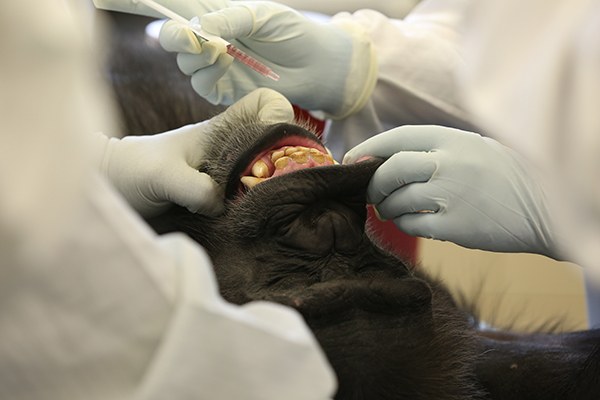
Researchers at the NIRC gave four chimpanzees an injection—and another six an oral dose—of a vaccine that consists of a virus-like protein, called a glycoprotein. It contains a small fragment of viral protein, but is incapable of replicating and causing infection.
After 28 days, the chimpanzees had high amounts of Ebola antibodies. “Everything indicates that if we take this to Africa and put it in chimps, it is going to be protective,” Walsh told Science.
The article is based on a paper recently published by Walsh and several other researchers in the journal Scientific Reports. Walsh is lead author of the paper, which is titled “The Final (Oral Ebola) Vaccine Trial on Captive Chimpanzees?”
Despite the promising trial, additional research and advances are hindered by federal regulations limiting biomedical research on chimpanzees, Walsh asserts.
Improvements to the vaccine, to prevent diminished effectiveness in the heat of tropical climates for example, would require further testing on captive animals.
That likely won’t happen after passage of a provision to the Endangered Species Act that went into effect in 2016. It precludes biomedical research on chimpanzees without a permit.
The new rule makes it too costly to maintain chimpanzee groups for research, according to Walsh.
Both humans and great apes stand to suffer.
In 2014, an outbreak of the Zaire strain of the Ebola virus in West Africa claimed the lives of about 1,600 people, according to statistics from the Centers for Disease Control and Prevention.
Wild apes have also been decimated by Ebola, which is one of the leading killers of gorillas and chimpanzees. The disease has wiped out up to one-third of the world’s populations, according to some estimates.
A hemorrhagic fever virus, Ebola has a high attack rate and high death rate. It is spread through contact and, in some human cases, by eating the tainted meat of wild animals.
To learn more, view the Science article or Walsh’s paper.
Photo: The Ebola vaccine was placed inside both cheeks and under the tongue of some sedated chimpanzees. Other chimpanzees received it by intramuscular injection.